Abstract
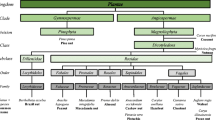
Radish is important as a root vegetable, a leafy vegetable, a fruit vegetable, an oil crop, and also as a cover plant. The economic importance and characteristics of radish differ between the East and the West of the world. In the East, there are radish cultivars having large roots with various shapes called “Asian big radish” and those grown for production of immature pods or oil seeds, whereas radish is a small vegetable grown within one month in the West. Asian big radish is expected to eventually become popular in the West. Radish belongs to the genus Raphanus, but is similar to the Brassica species except for the shape of pods and seeds. Despite their similarities, the order of genes in chromosomes is quite different between Raphanus and Brassica. Radish genome sequences have been published from three groups using similar cultivars, and therefore, collaboration for combining sequence data is considered to be effective for determination of more reliable genome sequences. Some radish lines have high salt tolerance and disease resistance different from Brassica crops. Radish also has a characteristic glucosinolate composition. Since radish can be crossed with Brassica species, it is also important as a genetic resource for Brassica crop breeding.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ahuja KL, Singh H, Raheja RK, Labana KS (1987) The oil content and fatty acid composition of various genotypes of cauliflower, turnip and radish. Plant Foods Hum Nutr 37:33–40
Akaba M, Kaneko Y, Hatakeyama K, Ishida M, Bang SW, Matsuzawa Y (2009) Identification and evaluation of clubroot resistance of radish chromosome using a Brassica napus–Raphanus sativus monosomic addition line. Breed Sci 59:203–206
Arumuganathan K, Earle ED (1991) Nuclear DNA content of some important species. Plant Mol Biol Rep 9:208–218
Barillari J, Cervellati R, Paolini M, Tatibouet A, Rollin P, Iori R (2005) Isolation of 4-methylthio-3-butenyl glucosinolate from Raphanus sativus sprouts (kaiware daikon) and its redox properties. J Agric Food Chem 53:9890–9896
Bennetzen J, Freeling M (1997) The unified grass genome: synergy in synteny. Genome Res 7:301–306
Cho MA, Min SR, Ko SM, Liu JR, Choi PS (2008) Agrobacterium-mediated genetic transformation of radish (Raphanus sativus L.). Plant Biotechnol 25:205–208
Gomez-Campo C (1980) Morphology and morpho-taxonomy of the tribe Brassiceae. In: Tsunoda S et al (eds) Brassca crops and wild allies. Japan Scientific Societies Press, Tokyo, pp 3–31
Harberd DJ, McArthur ED (1980) Meiotic analysis of some species and genus hybrids in the Brassiceae. In: Tsunoda S et al (eds) Brassca crops and wild allies. Japan Scientific Societies Press, Tokyo, pp 65–87
Hashida T, Nakatsuji R, Budahn H, Schrader O, Peterka H, Fujimura T, Kubo N, Hirai M (2013) Construction of a chromosome-assigned, sequence-tagged linkage map for the radish, Raphanus sativus L. and QTL analysis of morphological traits. Breed Sci 63:218–226
Inaba R, Nishio T (2002) Phylogenetic analysis of Brassiceae based on the nucleotide sequences of the S-locus related gene, SLR1. Theor Appl Genet 105:1159–1165
Ishida M, Kakizaki T, Morimitsu Y, Ohara T, Hatakeyama K, Yoshiaki H, Kohori J, Nishio T (2015) Novel glucosinolate composition lacking 4-methylthio-3-butenyl glucosinolate in Japanese white radish (Raphanus sativus L.). Thoer Appl Genet 128:2037–2046
Jeong Y-M, Kim N, Ahn B, Oh M, Chung W-H, Chung H, Jeong S, Lim K-B, Hwang Y-J, Kim G-B, Baek S, Choi S-B, Hyung D-J, Lee S-W, Sohn S-H, Kwon S-J, Jin M, Seol Y-J, Chae W, Choi K, Park B-S, Yu H-J, Mun J-H (2016) Elucidating the triplicated ancestral genome structure of radish based on chromosome-level comparison with the Brassica genomes. Theor Appl Genet 129:1357–1372
Johnston JS, Pepper AE, Hall AE, Chen ZJ, Hodnett G, Drabek J, Lopez R, Price HJ (2005) Evolution of genome size in Brassicaceae. Ann Bot 95:229–235
Kakizaki T, Kitashiba H, Zou Z, Li F, Fukino N, Ohara T, Nishio T, Ishida M (2017) A 2-oxoglutarate-dependent dioxygenase mediates the biosynthesis of glucoraphasatin in radish. Plant Physiol 173:1583–1593
Karpechenko GD (1924) Hybrids of Raphanus sativus L. x Brassica oleracea L. J Genet 14:375–396
Kamei A, Tsuro M, Kubo N, Hayashi T, Wang N, Fujimura T, Hirai M (2010) QTL mapping of clubroot resistance in radish (Raphanus sativus L.). Theor Appl Genet 120:1021–1027
Kitashiba H, Li F, Hirakawa H, Kawanabe T, Zou Z, Hasegawa Y, Tonosaki K, Shirasawa S, Fukushima A, Yokoi S, Takahata Y, Kakizaki T, Ishida M, Okamoto S, Sakamoto K, Shirasawa K, Tabata S, Nishio T (2014) Draft sequences of the radish (Raphanus sativus L.) genome. DNA Res 21:481–490
Koizuka N, Imai R, Fujimoto H, Hayakawa T, Kimura Y, Kohno-Murase J, Sakai T, Kawasaki S, Imamura J (2003) Genetic characterization of a pentatricopeptide repeat protein gene, orf587, that restores fertility in the cytoplasmic male-sterile Kosena radish. Plant J 34:407–415
Lelivelt CLC, Lang W, Dolstra O (1993) Intergeneric crosses for the transfer of resistance to the beet cyst nematode from Raphanus sativus to Brassica napus. Euphytica 58:111–120
Li F, Hasegawa Y, Saito M, Shirasawa S, Fukushima A, Ito T, Fujii H, Kishitani S, Kitashiba H, Nishio T (2011) Extensive chromosome homoeology among Brassiceae species were revealed by comparative genetic mapping with high-density EST-based SNP markers in radish (Raphanus sativus L.). DNA Res 18:401–411
Lim SH, Cho HJ, Lee SJ, Cho YH, Kim BD (2002) Identification and classification of S haplotypes in Raphanus sativus by PCR-RFLP of the S locus glycoprotein (SLG) gene and the S locus receptor kinase (SRK) gene. Theor Appl Genet 104:1253–1262
McCallum CM, Comai L, Greene EA, Henikoff S (2000) Targeted screening for induced mutations. Nat Biotechnol 18:455–457
Mitsui Y, Shimomura M, Komatsu K, Namiki N, Shibata-Hatta M, Imai M, Katayose Y, Mukai Y, Kanamori H, Kurita K, Kagami T, Wakatsuki A, Ohyanagi H, Ikawa H, Minaka N, Nakagawa K, Shiwa Y, Sasaki T (2015) The radish genome and comprehensive gene expression profile of tuberous root formation and development. Sci Rep 5:10835
Mizushima U (1980) Genome analysis in Brasica and allied genera. In: Tsunoda S et al (eds) Brassca crops and wild allies. Japan Scientific Societies Press, Tokyo, pp 89–106
Mun J-H, Chung H, Chung W-H, Oh M, Jeong Y-M, Kim N, Ahn BO, Park B-S, Park S, Lim K-B, Hwang Y-J, Yu H-J (2015) Construction of a reference genetic map of Raphanus sativus based on genotyping by whole-genome resequencing. Theor Appl Genet 128:259–272
Nasu S, Kitashiba H, Nishio T (2012) “Na-no-hana Project” for recovery from the Tsunami disaster by producing salinity-tolerant oilseed rape lines: Selection of salinity-tolerant lines of Brassica crops. J Integr Field Sci 9:33–37
Niikura S, Matsuura S (1998) Identification of self-incompatibility alleles (S) by PCR-RFLP in radish (Raphanus sativus L.). Euphytica 102:379–384
Niikura S, Matsuura S (2000) Genetic analysis of the reaction level of self-incompatibility to a 4% CO2 gas treatment in the radish (Raphanus sativus L.). Theor Appl Genet 101:1189–1193
Nishio T, Kusaba M, Watanabe M, Hinata K (1996) Registration of S alleles in Brassica campestris L by the restriction fragment sizes of SLGs. Theor Appl Genet 92:388–394
Okamoto S, Sato Y, Sakamoto K, Nishio T (2004) Distribution of similar self-incompatibility (S) haplotypes in different genera, Raphanus and Brassica. Sex Plant Reprod 17:33–39
Ozeki Y (2010) Study on the relationship between the anthocyanin molecular species and color phenotype of roots in the inbred lines of red radish (Raphanus sativus L.). Japan Food Chem Res Found Res Rep 16:33–39 (in Japanese with English summary)
Papi A, Orlandi M, Bartolini G, Barillari J, Iori R, Paolini M, Ferroni F, Fumo MG, Pedulli GF, Velgimigli L (2008) Cytotoxic and antioxidant activity of 4-methylthio-3-butenyl isothiocyanate from Raphanus sativus L. (kaiware daikon) sprouts. J Agric Food Chem 56:875–883
Park B-J, Liu Z, Kanno A, Kameya T (2005) Transformation of radish (Raphanus sativus L.) via sonication and vacuum infiltration of germinated seeds with Agrobacterium harboring a group 3 LEA gene from B. napus. Plant Cell Rep 24:494–500
Park NI, Xu H, Li X, Jang IH, Park S, Ahn GH, Lim YP, Kim SJ, Park SU (2011) Anthocyanin accumulation and expression of anthocyanin biosynthetic genes in radish (Raphanus sativus). J Agric Food Chem 59:6034–6039
Peters SA, Bargsten JW, Szinay D, van de Belt J, Visser RGF, Bai Y, de Jong H (2012) Structural homology in the Solanaceae: analysis of genomic regions in support of synteny studies in tomato, potato and pepper. Plant J 71:602–614
Sakamoto K, Kusaba M, Nishio T (1998) Polymorphism of the S-locus glycoprotein gene (SLG) and the S-locus related gene (SLR1) in Raphanus sativus L. and self-incompatible ornamental plants in the Brassicaceae. Mol Gen Genet 258:397–403
Shen D, Shu H, Huang M, Zheng Y, Li X, Fei Z (2013) RadishBase: a database for genomics and genetics of radish. Plant Cell Physiol 54(1–6):e3
Shirasawa K, Oyama M, Hirakawa H, Sato S, Tabata S, Fujioka T, Kimizuka-Takagi C, Sasamoto S, Watanabe A, Kato M, Kishida Y, Kohara M, Takahashi C, Tsuruoka H, Wada T, Sakai T, Isobe S (2011) An EST-SSR linkage map of Raphanus sativus and comparative genomics of the Brassicaceae. DNA Res 18:221–232
Shishu KIP (2009) Inhibition of cooked food-induced mutagenesis by dietary constituents: comparison of two natural isothiocyanates. Food Chem 112:977–981
Takahata Y, Komatsu H, Kaizuma N (1996) Microspore culture of radish (Raphanus sativus L.): influence of genotype and culture conditions on embryogenesis. Plant Cell Rep 16:163–166
Terasawa Y (1932) Tetraploid Bastarde von Brassica chinensis L. x Raphanus sativus L. Jpn J Genet 7:183–185 (In Japanese)
Tonosaki K, Michiba K, Bang SW, Kitashiba H, Kaneko Y, Nishio T (2013) Genetic analysis of hybrid seed formation ability of Brassica rapa in intergeneric crossings with Raphanus sativus. Theor Appl Genet 126:837–846
Tsuro M, Suwabe K, Kubo N, Matsumoto S, Hirai M (2008) Mapping of QTLs controlling root shape and red pigmentation in radish, Raphanus sativus L. Breed Sci 58:55–61
Warwick SI, Black LD (1997) Phylogenetic implications of chloroplast DNA restriction site variation in subtribes Raphaninae and Cakilinae (Brassicaceae, tribe Brassiceae). Can J Bot 75:960–973
Warwick SI, Hall JC (2009) Phylogeny of Brassica and wild relatives. In: Gupta SK (ed) Biology and breeding of crucifers. CRC Press, London, pp 19–36
Warwick SI, Sauder CA (2005) Phylogeny of tribe Brassiceae (Brassicaceae) based on chloroplast restriction site polymorphisms and nuclear ribosomal internal transcribed spacer and chloroplast trnL intron sequences. Can J Bot 83:467–483
Weil RR, Kremen A (2007) Thinking across and beyond disciplines to make cover crops pay. J Sci Food Agric 87:551–557
Yamagishi H, Terachi T (1996) Molecular and biological studies on male-sterile cytoplasm in the Cruciferae III. Distributtion of Ogura-type cytoplasm among Japanese wild radishes and Asian radish cultivars. Theor Appl Genet 93:325–332
Zou Z, Ishida M, Li F, Kakizaki T, Suzuki S, Kitashiba H, Nishio T (2013) QTL analysis using SNP markers developed by next-generation sequencing for identification of candidate genes controlling 4-methylthio-3-butenyl glucosinolate contents in roots of radish, Raphanus sativus L. PLoS ONE 8:e53541
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Nishio, T. (2017). Economic and Academic Importance of Radish. In: Nishio, T., Kitashiba, H. (eds) The Radish Genome. Compendium of Plant Genomes. Springer, Cham. https://doi.org/10.1007/978-3-319-59253-4_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-59253-4_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-59252-7
Online ISBN: 978-3-319-59253-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)