Abstract
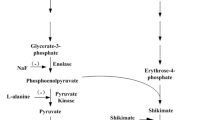
To study the effect of DNA methylation on the growth and synthesis of cephalotaxine in Cephalotaxus mannii suspension cells, demethylating reagent 5-aza-2′-deoxycytidine (5-Aza-CdR) with different concentrations (15, 25 and 35 μmol/L) was added to the suspension cultures of C. mannii. The results obtained revealed that the effect of 5-Aza-CdR on cell growth depended on its concentration. When cells were treated with an optimum concentration of 5-Aza-CdR 15 μmol/L, the biomass of cells was 15.76 g/L (dry weight, DW), 13.75% higher than control group (13.85 g/L). Additionally, 5-Aza-CdR could improve the enzyme activity associated with the metabolic pathway of cell growth and product biosynthesis. When treated with 5-Aza-CdR, the activities of glucose-6-phosphate dehydrogenase (G6PDH), pyruvate kinase (PK) and 3-deoxy-d-arabino-heptulosonate-7-phosphate (DS) synthase was increased by 21%, 172% and 26%, respectively. At the same time, the content of cephalotaxine reached 7.735 mg/L, was 30% higher than the control group (5.97 mg/L). In addition, it was found that 5-Aza-CdR reduced the expression of two genes related to methyltransferase in cells by 87.87% (caffeic acid ortho-methyltransferase gene) and 86.19% (one hypothetical protein gene), and the level of DNA methylation treated by 5-Aza-CdR was obviously lower than the control. Interestingly, the demethylation by 5-Aza-CdR was only found in the cell growth phase (days 0–15), but not found in the late culture period (days 15–30), indicating that the action sites of 5-Aza-CdR in cells were mainly focused on the primary metabolic process.
Key message
5-Aza-CdR effected the prophase of cell growth and promoted the synthesis of cephalotaxine. The decrease of DNA methylation level by 5-Aza-CdR was only found in the cell growth phase (0–15 days). But there had no significant effect at the later stage (15–30 days) of culture.







Similar content being viewed by others
Abbreviations
- 5-Aza-CdR:
-
5-Aza-2′-deoxycytidine
- G6PDH:
-
Glucose-6-phosphate dehydrogenase
- PK:
-
Pyruvate kinase
- DS:
-
3-Deoxy-d-arabino-heptulosonate-7-phosphate synthase
- SAM:
-
Methyl methionine
- MBD:
-
Methyl binding protein
- NAA:
-
Naphthalene acetic acid
- KT:
-
Kinetin
- PVP:
-
Polyvinyl pyrrolidone
- FW:
-
Fresh weight
- DW:
-
Dry weight
- HPLC:
-
High performance liquid chromatography
- SDS:
-
Sodium dodecyl sulfate
- EDTA:
-
Ethylene diamine tetraacetic acid
- LDH:
-
Lactic dehydrogenase (rabbit muscle)
- E4P:
-
Erythrose-4-phosphate
- PEP:
-
Phosphoenolpyruvate
References
Akimoto K, Katakami H, Kim HJ, Ogawa E, Sano CM, Wada Y, Sano H (2007) Epigenetic inheritance in rice plants. Ann Bot 100(2):205–217
Baubec T, Pecinka A, Rozhon W, Scheid OM (2009) Effective, homogeneous and transient interference with cytosine methylation in plant genomic DNA by zebularine. Plant J Cell Mol Biol 57(3):542–554
Bartels A, Han Q, Nair P, Stacey L, Gaynier H, Mosley M, Huang Q, Pearson J, Hsieh TF, An YQ, Xiao W (2018) Dynamic DNA methylation in plant growth and development. Int J Mol Sci 19(7):2144
Bird A (2002) DNA methylation patterns and epigenetic memory. Genes Dev 16(1):6–21
Bouyer D, Kramdi A, Kassam M, Heese M, Schnittger A, Roudier F, Colot V (2017) DNA methylation dynamics during early plant life. Genome Biol 18(1):179
Demeulemeester MAC, Stallen NV, Proft MPD (1999) Degree of DNA methylation in chicory (Cichorium intybus L.): influence of plant age and vernalization. Plant Sci 142(1):101–108
Finnegan EJ, Peacock WJ, Dennis ES (2000) DNA methylation, a key regulator of plant development and other processes. Curr Opin Genet Dev 10(2):217–223
Fransz PF, Jong JHD (2002) Chromatin dynamics in plants. Curr Opin Plant Biol 5(6):560–567
Fu C, Li L, Wu W, Li M, Yu X, Yu L (2012) Assessment of genetic and epigenetic variation during long-term Taxus cell culture. Plant Cell Rep 31(7):1321–1331
Gao Z, Liu HL, Daxinger L, Pontes O, He X, Qian W, Lin HX, Xie MT, Lorkovic ZJ, Zhang SD, Miki D, Zhan XQ, Pontier D, Lagrange T, Jin HL, Matzke AJM, Matzke M, Pikaard CS, Zhu JK (2010) An RNA polymerase II- and AGO4-associated protein acts in RNA-directed DNA methylation. Nature 465(7294):106–109
Gong Z, Morales-Ruiz T, Ariza RR, Roldán-Arjona T, David L, Zhu JK (2002) ROS1, a repressor of transcriptional gene silencing in Arabidopsis, encodes a DNA glycosylase/lyase. Cell 111(6):803–814
Gundry M, Vijg J (2012) Direct mutation analysis by high-throughput sequencing: from germline to low-abundant, somatic variants. Mutat Res 729(1–2):1–15
Haaf T (1995) The effects of 5-azacytidine and 5-azadeoxycytidine on chromosome structure and function: implications for methylation-associated cellular processes. Pharmacol Ther 65(1):19–46
Huang SS, Ecker J (2017) Piecing together, cis-regulatory networks: insights from epigenomics studies in plants. Wiley Interdiscip Rev Syst Biol Med 10(3):e1411
Huang LC, Hsiao LJ, Pu SY, Kuo CI, Huang BL, Tseng TC, Huang HJ, Chen YT (2012) DNA methylation and genome rearrangement characteristics of phase change in cultured shoots of Sequoia sempervirens. Physiol Plant 145(2):360–368
Jones PA (1985) Altering gene expression with 5-azacytidine. Cell 40(3):485–486
Jullien PE, Mosquna A, Ingouff M, Sakata T, Ohad N, Berger F (2008) Retinoblastoma and its binding partner MSI1 control imprinting in Arabidopsis. PLoS Biol 6(8):1693–1705
Kiselev KV, Tyunin AP, Manyakhin AY, Zhuravlev YN (2011) Resveratrol content and expression patterns of stilbene synthase genes in Vitis amurensis cells treated with 5-azacytidine. Plant Cell Tissue Organ Cult 105(1):65–72
Kiselev KV, Dubrovina AS, Shumakova OA (2013) DNA mutagenesis in 2- and 20-yr-old Panax ginseng cell cultures. Vitro Cell Dev Biol - Plant 49(2):175–182
Kwiatkowska A, Zebrowski J, Oklejewicz B, Czarnik J, Halibart-Puzio J, Wnuk M (2014) The age-dependent epigenetic and physiological changes in an Arabidopsis T87 cell suspension culture during long-term cultivation. Biochem Biophys Res Commun 447(2):285–291
Law RD, Suttle JC (2005) Chromatin remodeling in plant cell culture: patterns of DNA methylation and histone H3 and H4 acetylation vary during growth of asynchronous potato cell suspensions. Plant Physiol Biochem (Paris) 43(6):527–534
Li YC (2014) Enhanced cephalotaxine production in Cephalotaxus mannii suspension cultures by combining glycometabolic regulation and elicitation. Process Biochem 49(12):2279–2284
Li YC, Jiang XX (2019) Improving of cephalotaxine production in Cephalotaxus mannii suspension cultures by alanine and its derivatives. Plant Cell Tissue Organ Cult 136(1):101–108
Li D, Yu X, Jiang X, Wang L, Wang F (2010) Effects of 5-azaC on vernalization and flower-ball development in broccoli (Brassica oleracea var. italica). Pak J Bot 42(3):1889–1893
Li YC, Jiang XX, Long XJ (2014) Effects and action mechanisms of sodium fluoride (NaF) on the growth and cephalotaxine production of Cephalotaxus mannii suspension cells. Enzyme Microbial Technol 67:77–81
Liu T, Li Y, Duan W, Huang F, Hou X (2017) Cold acclimation alters DNA methylation patterns and confers tolerance to heat and increases growth rate in Brassica rapa. J Exp Bot 68(5):1213–1224
Mager S, Ludewig U (2018) Massive loss of DNA methylation in nitrogen-, but not in phosphorus-deficient zea mays roots is poorly correlated with gene expression differences. Front Plant Sci 9:497
Mankessi F, Saya AR, Favreau B, Doulbeau S, Conéjéro G, Lartaud M, Verdeil JL, Monteuuis O (2011) Variations of DNA methylation in Eucalyptus urophylla × Eucalyptus grandis shoot tips and apical meristems of different physiological ages. Physiol Plant 143(2):178–187
Matasyoh LG, Wachira FN, Kinyua MG, Muigai A, Mukiama TK (2008) Leaf storage conditions and genomic DNA isolation efficiency in Ocimum gratissimum L. from Kenya. Afr J Biotechnol 7(5):557–564
Mccandliss RJ, Poling MD, Herrmann KM (1978) 3-Deoxy-D-arabino-heptulosonate 7-phosphate synthase. Purification and molecular characterization of the phenylalanine-sensitive isoenzyme from Escherichia coli. J Biol Chem 253(12):4259–4265
Moarefi AH, Frédéric C (2011) ICF syndrome mutations cause a broad spectrum of biochemical defects in DNMT3B-mediated de novo DNA methylation. J Mol Biol 409(5):758–772
Podestá FE, Plaxton WC (1992) Plant cytosolic pyruvate kinase: a kinetic study. Biochim Biophys Acta (BBA) 1160(2):213–220
Powell RG (1979) Cephalomannine; a new antitumour alkaloid from Cephalotaxus mannii. J Chem Soc Chem Commun 3(3):102–104
Rosati VC, Quinn AA, Fromhold SM, Gleadow R, Blomstedt CK (2019) Investigation into the role of DNA methylation in cyanogenesis in sorghum (Sorghum bicolor L. Moench). Plant Growth Regul 88(1):73–85
Seelan RS, Mukhopadhyay P, Pisano MM, Greene RM (2018) Effects of 5-aza-2′-deoxycytidine (decitabine) on gene expression. Drug Metab Rev 50(2):1–15
Song Y, Liu L, Li G, An L, Tian L (2017) Trichostatin A and 5-Aza-2’-Deoxycytidine influence the expression of cold-induced genes in Arabidopsis. Plant Signal Behav 12(2):e1389828
Takano I, Yasuda I, Nishijima M, Hitotsuyanagi Y, Takeya K, Itokawa H (1996) Alkaloids from Cephalotaxus harringtonia. Phytochemistry 43(1):299–303
Tatra GS, Miranda J, Chinnappa CC, Reid DM (2010) Effect of light quality and 5-azacytidine on genomic methylation and stem elongation in two ecotypes of Stellaria longipes. Physiol Plant 109(3):313–321
Tyunin AP, Kiselev KV, Zhuravlev YN (2012) Effects of 5-azacytidine induced DNA demethylation on methyltransferase gene expression and resveratrol production in cell cultures of Vitis amurensis. Plant Cell Tissue Organ Cult 111(1):91–100
Victoria D, Aliki K, Venetia K, Georgios M, Zoe H (2017) Spatial and temporal expression of cytosine-5 DNA methyltransferase and DNA demethylase gene families of the Ricinus communis during seed development and drought stress. Plant Growth Regul 84(3):1–14
Xia J (2008) Study on the factors effect on stable production of taxol in Taxus cells. Huazhong University of Science and Technology
Yu LJ, Lan WZ, Chen C, Yang Y (2004) Glutathione levels control glucose-6-phosphate dehydrogenase activity during elicitor-induced oxidative stress in cell suspension cultures of Taxus chinensis. Plant Sci 167(2):329–335
Zhang H, Zhu JK (2012) Active DNA demethylation in plants and animals. Cold Spring Harb Symp Quant Biol 77(4293):161–173
Zhang W, Curtin C, Franco C (2002) Towards manipulation of post-biosynthetic events in secondary metabolism of plant cell cultures. Enzyme Microbial Technol 30(6):688–696
Zhang M, Kimatu JN, Xu K, Liu B (2010) DNA cytosine methylation in plant development. J Genet Genom 37(1):1–12
Zhang H, Lang Z, Zhu JK (2018) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19(1):489–506
Acknowledgements
This work was funded by the National Natural Science Foundation of China (No. 21766006).
Author information
Authors and Affiliations
Contributions
LRW and YCL conceived and designed the study. LRW conducted experiments, analyzed the results and wrote the manuscripts. YCL and WYQ revised the manuscript in part. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest to this work.
Research involving human and animal participants
No human participants and/or animals are involved in this work.
Additional information
Communicated by Konstantin V. Kiselev.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wei, LR., Qin, WY. & Li, YC. Effects of demethylating reagent 5-aza-2′-deoxycytidine on the growth and cephalotaxine production in Cephalotaxus mannii suspension cells. Plant Cell Tiss Organ Cult 139, 359–368 (2019). https://doi.org/10.1007/s11240-019-01689-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01689-1