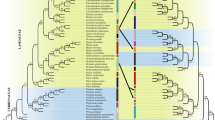
Abstract
Recently developed computational tools in ITS2 sequence-structure phylogenetics are improving tree robustness by exploitation of the added information content of the secondary structure. Despite this strength, however, their adoption for species-level clarifications in angiosperms has been slow. We investigate the utility of combining ITS2 sequence and secondary structure to separate species of southern African Strychnos, and assess correlation between compensatory base changes (CBCs) and currently recognised species boundaries. Combined phylogenetic analysis of sequence and secondary structure datasets performed better, in terms of robustness and species resolution, than analysis involving primary sequences only, achieving 100 and 88.2 % taxa discriminations respectively. Further, the Strychnos madagascariensis complex is well-resolved by sequence-structure phylogenetic analysis. The 17 Strychnos species corresponded to 14 ITS2 CBC clades. Four of the five taxa in section Densiflorae belong to a single CBC clade, whose members tend to form natural hybrids. Our finding supports the application of ITS2 as a complementary barcoding marker for species identification. It also highlights the potential of comparative studies of ITS2 CBC features among prospective parental pairs in breeding experiments as a rapid proxy for cross compatibility assessment. This could save valuable time in crop improvement. Patterns of CBC evolution and species boundaries in Strychnos suggests a positive correlation. We conclude that the CBC pattern coupled with observed ITS2 sequence paraphyly in section Densiflorae points to a speciation work-in-progress.




Similar content being viewed by others
References
Adams DC, Berns CM, Kozak KH, Wiens JJ (2009) Are rates of species diversification correlated with rates of morphological evolution? Proc R Soc Lond 276:2729–2738
Adebowale A (2014) Biosystematic studies in southern African species of Strychnos L. (Loganiaceae). Doctoral dissertation, University of KwaZulu-Natal, Durban
Adebowale A, Nicholas A, Lamb J, Naidoo Y (2012) Elliptic Fourier analysis of leaf shape in southern African Strychnos section Densiflorae (Loganiaceae). Bot J Linn Soc 170:542–553
Adebowale A, Lamb J, Nicholas A, Naidoo Y (2016) Molecular systematics of southern African monkey orange Strychnos L. (Loganiaceae). Kew Bull 71:1–16
Alvarez I, Wendel JF (2003) Ribosomal ITS sequences and plant phylogenetic inference. Mol Phylogenet Evol 29:417–434
Ankenbrand MJ, Keller A, Wolf M, Schultz J, Forster F (2015) ITS2 database V: twice as much. Mol Biol Evol 32:3030–3032
Baldwin BG, Sanderson MJ, Porter JM, Wojciechowski MF, Campbell CS, Donoghue MJ (1995) The ITS region of nuclear ribosomal DNA: a valuable source of evidence on angiosperm phylogeny. Ann Mo Bot Gard 82:247–277
Biswal DK, Debnath M, Kumar S, Tandon P (2012) Phylogenetic reconstruction in the Order Nymphaeales: ITS2 secondary structure analysis and in silico testing of maturase k (matK) as a potential marker for DNA bar coding. BMC Bioinform 13:S26–S26
Bottin L, Le Cadre S, Quilichini A, Bardin P, Moret J, Machon N (2007) Re-establishment trials in endangered plants: a review and the example of Arenaria grandiflora, a species on the brink of extinction in the Parisian region (France). Ecoscience 14:410–419
Buchheim MA, Keller A, Koetschan C, Forster F, Merget B, Wolf M (2011) Internal transcribed spacer 2 (nu ITS2 rRNA) sequence-structure phylogenetics: towards an automated reconstruction of the green algal tree of life. PLoS ONE 6:e16931
Caisova L, Marin B, Melkonian M (2011) A close-up view on ITS2 evolution and speciation—a case study in the Ulvophyceae (Chlorophyta, Viridiplantae). BMC Evol Biol 11:262
CBOL Plant Working Group (2009) A DNA barcode for land plants. Proc Natl Acad Sci USA 106:12794–12797
Chen S, Yao H, Han J, Liu C, Song J, Shi L, Zhu Y, Ma X, Gao T, Pang X, Luo K, Li Y, Li X, Jia X, Lin Y, Leon C (2010) Validation of the ITS2 region as a novel DNA barcode for identifying medicinal plant species. PLoS One 5:e8613
Coleman AW (2000) The significance of a coincidence between evolutionary landmarks found in mating affinity and a DNA sequence. Protist 151:1–9
Coleman AW (2003) ITS2 is a double-edged tool for eukaryote evolutionary comparisons. Trends Genet 19:370–375
Coleman AW (2007) Pan-eukaryote ITS2 homologies revealed by RNA secondary structure. Nucleic Acids Res 35:3322–3329
Coleman AW (2009) Is there a molecular key to the level of “biological species” in eukaryotes? A DNA guide. Mol Phylogenet Evol 50:197–203
del Campo EM, Catala S, Gimeno J, del Hoyo A, Martinez-Alberola F, Casano LM, Grube M, Barreno E (2013) The genetic structure of the cosmopolitan three-partner lichen Ramalina farinacea evidences the concerted diversification of symbionts. FEMS Microbiol Ecol 83:310–323
Draisma SA, Eurlings MM, Lim P-E (2012) High intra-individual sequence variation in the nuclear rDNA LSU-5S intergenic spacer in the Sargassaceae (Fucales, Phaeophyceae). J Appl Phycol 24:1373–1379
Eddy SR (1998) Profile hidden Markov models. Bioinformatics 14:755–763
Edger PP, Tang M, Bird KA, Mayfield DR, Conant G, Mummenhoff K, Koch MA, Pires JC (2014) Secondary structure analyses of the nuclear rRNA internal transcribed spacers and assessment of its phylogenetic utility across the Brassicaceae (Mustards). PloS One 9:e101341
Frankham R, Ballou JD, Dudash MR, Eldridge MDB, Fenster CB, Lacy RC, Mendelson JR III, Porton IJ, Ralls K, Ryder OA (2012) Implications of different species concepts for conserving biodiversity. Biol Conserv 153:25–31
Frasier LC (2008) Evolution and systematics of the angiosperm order Gentianales with an in-depth focus on Loganiaceae and its species-rich and toxic genus Strychnos. Doctoral dissertation. Rutgers, The State University of New Jersey, New Jersey
Gao T, Yao H, Song J, Zhu Y, Liu C, Chen S (2010) Evaluating the feasibility of using candidate DNA barcodes in discriminating species of the large Asteraceae family. BMC Evol Biol 10:324
Gascuel O (1997) BIONJ: an improved version of the NJ algorithm based on a simple model of sequence data. Mol Biol Evol 14:685–695
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Gutell RR, Larsen N, Woese CR (1994) Lessons from an evolving rRNA: 16S and 23S rRNA structures from a comparative perspective. Microbiol Rev 58:10–26
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Harpke D, Peterson A (2006) Non-concerted ITS evolution in Mammillaria (Cactaceae). Mol Phylogenet Evol 41:579–593
Hebert PD, Cywinska A, Ball SL, deWaard JR (2003) Biological identifications through DNA barcodes. Proc Biol Sci 270:313–321
Hershkovitz MA, Zimmer EA (1996) Conservation patterns in angiosperm rDNA ITS2 sequences. Nucleic Acids Res 24:2857–2867
Hodkinson TR, Chase MW, Lledó DM, Salamin N, Renvoize SA (2002) Phylogenetics of Miscanthus, Saccharum and related genera (Saccharinae, Andropogoneae, Poaceae) based on DNA sequences from ITS nuclear ribosomal DNA and plastid trnL intron and trnL-F intergenic spacers. J Plant Res 115:381–392
Hollingsworth PM (2011) Refining the DNA barcode for land plants. Proc Natl Acad Sci USA 108:19451–19452
Hollingsworth PM, Graham SW, Little DP (2011) Choosing and using a plant DNA barcode. PLoS One 6:e19254
Hughes CE, Bailey CD, Harris SA (2002) Divergent and reticulate species relationships in Leucaena (Fabaceae) inferred from multiple data sources: insights into polyploid origins and nrDNA polymorphism. Am J Bot 89:1057–1073
Hunter RL, LaJeunesse TC, Santos SR (2007) Structure and evolution of the rdna internal transcribed spacer (ITS) region 2 in the symbiotic dinoflagellates (Symbiodinium, Dinophyta) 1. J Phycol 43:120128
Inagaki MN, Tahir M (1992) Production of haploid wheat through intergeneric crosses. Hereditas 116:117–120
Inkscape. In., 0.48 edn; https://inkscape.org/en/ Accessed 12 Mar 2015
IUCN (2016) Red list of threatened species. http://www.iucnredlist.org. Accessed 25 July 2016
Joseph N, Krauskopf E, Vera MI, Michot B (1999) Ribosomal internal transcribed spacer 2 (ITS2) exhibits a common core of secondary structure in vertebrates and yeast. Nucleic Acids Res 27:4533–4540
Kaneko Y, Bang SW (2014) Interspecific and intergeneric hybridization and chromosomal engineering of Brassicaceae crops. Breed Sci 64:14–22
Keller A, Schleicher T, Förster F, Ruderisch B, Dandekar T, Müller T, Wolf M (2008) ITS2 data corroborate a monophyletic chlorophycean DO-group (Sphaeropleales). BMC Evol Biol 8:218
Keller A, Schleicher T, Schultz J, Muller T, Dandekar T, Wolf M (2009) 5.8S-28S rRNA interaction and HMM-based ITS2 annotation. Gene 430:50–57
Keller A, Forster F, Muller T, Dandekar T, Schultz J, Wolf M (2010) Including RNA secondary structures improves accuracy and robustness in reconstruction of phylogenetic trees. Biol Direct 5:4
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kocot KM, Santos SR (2009) Secondary structural modeling of the second internal transcribed spacer (ITS2) from Pfiesteria-like dinoflagellates (Dinophyceae). Harmful Algae 8:441–446
Koetschan C, Forster F, Keller A, Schleicher T, Ruderisch B, Schwarz R, Muller T, Wolf M, Schultz J (2010) The ITS2 database III—sequences and structures for phylogeny. Nucleic Acids Res 38:D275–D279
Koetschan C, Hackl T, Muller T, Wolf M, Forster F, Schultz J (2012) ITS2 database IV: interactive taxon sampling for internal transcribed spacer 2 based phylogenies. Mol Phylogenet Evol 63:585–588
Leeuwenberg AJM (1969) The Loganiaceae of Africa VIII. Strychnos III: revision of the African species with notes on the extra-African. Mededel Landbouwhogeschool Wageningen 69:1–316
Mai JC, Coleman AW (1997) The internal transcribed spacer 2 exhibits a common secondary structure in green algae and flowering plants. J Mol Evol 44:258–271
Mathews DH, Disney MD, Childs JL, Schroeder SJ, Zuker M, Turner DH (2004) Incorporating chemical modification constraints into a dynamic programming algorithm for prediction of RNA secondary structure. Proc Natl Acad Sci USA 101:7287–7292
Mayr E (1982) The growth of biological thought: diversity, evolution and inheritance. Belknap, Cambridge
Merget B, Wolf M (2010) A molecular phylogeny of Hypnales (Bryophyta) inferred from ITS2 sequence-structure data. BMC Res Notes 3:1
Mizrahi Y, Nerd A, Sitrit Y (2002) New fruits for arid climates. In: Janick J, Whipkey A (eds) Trends in new crops and new uses. ASHS Press, Alexandria, pp 378–384
Molina J, Struwe L (2009) Utility of secondary structure in phylogenetic reconstructions using nrDNA ITS sequences: an example from Potalieae (Gentianaceae: Asteridae). Syst Bot 34:414–428
Müller T, Philippi N, Dandekar T, Schultz J, Wolf M (2007) Distinguishing species. RNA 13:1469–1472
Mwamba CK (2006) Monkey Orange. University of Southampton International Centre for Underutilised Crops, Southampton UK, Strychnos cocculoides
Nieto FG, Rossello JA (2007) Better the devil you know? Guidelines for insightful utilization of nrDNA ITS in species-level evolutionary studies in plants. Mol Phylogenet Evol 44:911–919
Padial JM, Miralles A, De la Riva I, Vences M (2010) The integrative future of taxonomy. Front Zool 7:16
Page RDM (1996) TREEVIEW: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Ponce-Gordo F, Fonseca-Salamanca F, Martinez-Diaz RA (2011) Genetic heterogeneity in internal transcribed spacer genes of Balantidium coli (Litostomatea, Ciliophora). Protist 162:774–794
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Reuter JS, Mathews DH (2010) RNAstructure: software for RNA secondary structure prediction and analysis. BMC Bioinformatics 11:129
Roca AL, Georgiadis N, Pecon-Slattery J, O’Brien SJ (2001) Genetic evidence for two species of elephant in Africa. Science 293:1473–1477
Rogers S, Bendich A (1987) Ribosomal RNA genes in plants: variability in copy number and in the intergenic spacer. Plant Mol Biol 9:509–520
Ruhl MW, Wolf M, Jenkins TM (2010) Compensatory base changes illuminate morphologically difficult taxonomy. Mol Phylogenet Evol 54:664–669
Sanguila MB, Siler CD, Diesmos AC, Nuneza O, Brown RM (2011) Phylogeography, geographic structure, genetic variation, and potential species boundaries in Philippine slender toads. Mol Phylogenet Evol 61:333–350
Schill R, Förster F, Dandekar T, Wolf M (2010) Using compensatory base change analysis of internal transcribed spacer 2 secondary structures to identify three new species in Paramacrobiotus (Tardigrada). Org Divers Evol 10:287–296
Schlick-Steiner BC, Steiner FM, Seifert B, Stauffer C, Christian E, Crozier RH (2010) Integrative taxonomy: a multisource approach to exploring biodiversity. Annu Rev Entomol 55:421–438
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, Chen W, Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci USA 109:6241–6246
Schultz J, Maisel S, Gerlach D, Muller T, Wolf M (2005) A common core of secondary structure of the internal transcribed spacer 2 (ITS2) throughout the Eukaryota. RNA 11:361–364
Schultz J, Muller T, Achtziger M, Seibel PN, Dandekar T, Wolf M (2006) The internal transcribed spacer 2 database—a web server for (not only) low level phylogenetic analyses. Nucleic Acids Res 34:W704–W707
Seibel P, Müller T, Dandekar T, Schultz J, Wolf M (2006) 4SALE—a tool for synchronous RNA sequence and secondary structure alignment and editing. BMC Bioinform 7:1–7
Seibel PN, Muller T, Dandekar T, Wolf M (2008) Synchronous visual analysis and editing of RNA sequence and secondary structure alignments using 4SALE. BMC Res Notes 1:91
Selig C, Wolf M, Muller T, Dandekar T, Schultz J (2008) The ITS2 database II: homology modelling RNA structure for molecular systematics. Nucleic Acids Res 36:D377–D380
Song J, Shi L, Li D, Sun Y, Niu Y, Chen Z, Luo H, Pang X, Sun Z, Liu C, Lv A, Deng Y, Larson-Rabin Z, Wilkinson M, Chen S (2012) Extensive pyrosequencing reveals frequent intra-genomic variations of internal transcribed spacer regions of nuclear ribosomal DNA. PLoS One 7:e43971
Swofford DL (2002) PAUP*. Phylogenetic analysis using parsimony (*and other methods). In., 4.0b10 edn. Sinauer Associates, Sunderland
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tippery NP, Les DH, Crawford DJ (2015) Evaluation of phylogenetic relationships in Lemnaceae using nuclear ribosomal data. Plant Biol 17:50–58
Trizzino M, Audisio P, Antonini G, De Biase A, Mancini E (2009) Comparative analysis of sequences and secondary structures of the rRNA internal transcribed spacer 2 (ITS2) in pollen beetles of the subfamily Meligethinae (Coleoptera, Nitidulidae): potential use of slippage-derived sequences in molecular systematics. Mol Phylogenet Evol 51:215–226
Tychonievich J, Warner RM (2011) Interspecific crossability of selected Salvia species and potential use for crop improvement. J Am Soc Hort Sci 136:41–47
Verdoorn IC (1963) Loganiaceae. Flora of southern. Africa 26:134–149
Vollmer SV, Palumbi SR (2004) Testing the utility of internally transcribed spacer sequences in coral phylogenetics. Mol Ecol 13:2763–2772
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribososmal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols. Academic Press, San Diego, pp 315–324
Wolf M (2015) ITS so much more. Trends Genet 31:175–176
Wolf M, Achtziger M, Schultz J, Dandekar T, Müller T (2005) Homology modeling revealed more than 20,000 rRNA internal transcribed spacer 2 (ITS2) secondary structures. RNA 11:1616–1623
Wolf M, Ruderisch B, Dandekar T, Schultz J, Muller T (2008) ProfDistS: (profile-) distance based phylogeny on sequence—structure alignments. Bioinformatics 24:2401–2402
Wolf M, Chen S, Song J, Ankenbrand M, Muller T (2013) Compensatory base changes in ITS2 secondary structures correlate with the biological species concept despite intragenomic variability in ITS2 sequences—a proof of concept. PLoS One 8:e66726
Wolf M, Koetschan C, Müller T (2014) ITS2, 18S, 16S or any other RNA—simply aligning sequences and their individual secondary structures simultaneously by an automatic approach. Gene 546:145–149
Xiao LQ, Moller M, Zhu H (2010) High nrDNA ITS polymorphism in the ancient extant seed plant Cycas: incomplete concerted evolution and the origin of pseudogenes. Mol Phylogenet Evol 55:168–177
Yao H, Song J, Liu C, Luo K, Han J, Li Y, Pang X, Xu H, Zhu Y, Xiao P, Chen S (2010) Use of ITS2 region as the universal DNA barcode for plants and animals. PLoS One 5:e13102
Yi Z, Song W, Gong J, Warren A, Al-Rasheid KAS, Al-Arifi S, Al-Khedhairy AA (2009) Phylogeny of six oligohymenophoreans (Protozoa, Ciliophora) inferred from small subunit rRNA gene sequences. Zool Scr 38:323–331
Zamir D (2001) Improving plant breeding with exotic genetic libraries. Nat Rev Genet 2:983–989
Acknowledgments
This work was supported by the NRF-Thuthuka grant awarded to the first author through YN. The authors thank Masego Kruger-Gaadingwe and David Styles for help with collection of Strychnos specimens, and the curator of the National Herbarium Pretoria for access to materials. We also thank eThekwini Municipality for collection access to conservation areas around Durban.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Comparison of ITS2 helices II and III across southern African Strychnos. Key: ang = S. angolensis; coc = S. cocculoides; dec = S. decussata; dip = S. diplotricha; ger = S. gerrardii; hen = S. henningsii; inn = S. innocua; luc = S. lucens; mad = S. madagascariensis; mel = S. mellodora; mit = S. mitis; pan = S. panganensis; pot = S. potatorum; pun = S. pungens; spi = S. spinosa; usa = S. usambarensis; xan = S. xantha (EPS 1865 kb)
Figure S2
Comparison of ITS2 helices I and IV across southern African Strychnos. Key: ang = S. angolensis; coc = S. cocculoides; dec = S. decussata; dip = S. diplotricha; ger = S. gerrardii; hen = S. henningsii; inn = S. innocua; luc = S. lucens; mad = S. madagascariensis; mel = S. mellodora; mit = S. mitis; pan = S. panganensis; pot = S. potatorum; pun = S. pungens; spi = S. spinosa; usa = S. usambarensis; xan = S. xantha. The ITS2 sequences identified as (GenBank: JF937984.1) and S. panganensis are incomplete. They lack helix IV. Note the identical sequences of S. angolensis and GenBank: JF937984.1, a possible misidentification as S. henningsii by Frasier (2008), even in the highly variable terminal loop. (EPS 1290 kb)
Rights and permissions
About this article
Cite this article
Adebowale, A., Lamb, J., Nicholas, A. et al. ITS2 secondary structure for species circumscription: case study in southern African Strychnos L. (Loganiaceae). Genetica 144, 639–650 (2016). https://doi.org/10.1007/s10709-016-9931-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-016-9931-0